BEIJING, Feb. 8 (Xinhua) - China has fast-tracked the approval of the testing kits for novel coronavirus pneumonia, an official with the National Medical Products Administration said Saturday.
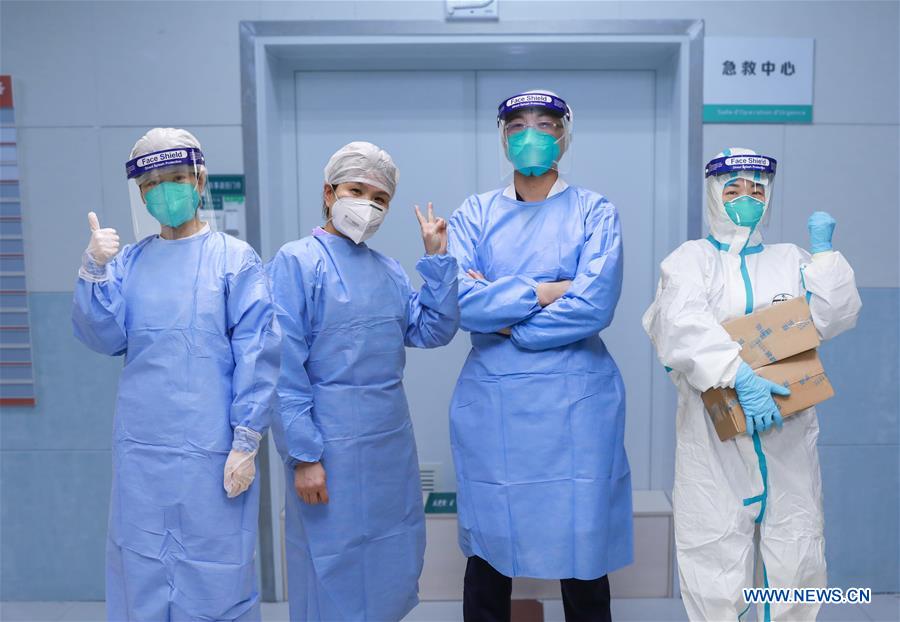
Health workers (L-R) Bai Hui, Li Chunfang, Zhao Zhigang and Guo Qin, who have recovered from recent novel coronavirus infections, pose for a group photo at the emergency medical center of Zhongnan Hospital of Wuhan University in Wuhan, central China's Hubei Province, Feb. 6, 2020. The four had been infected with the novel coronavirus during work. They will return to the healthcare frontline to continue battling the novel coronavirus epidemic. (Photo: Xinhua)
The registration of seven testing products from seven enterprises have been approved by the administration, expanding the supply capacity of nucleic acid testing kits, Jiang Deyuan, an official with the administration, said at a press conference in Beijing.
The performance and safety of these products have met the technical requirements after strict testing, he added.
The administration has required provincial medical products departments to fast-track approval of all kinds of medical equipment for epidemic prevention, Jiang said.
He pointed out that a total of 88 applications for medical equipment registration had been approved following the procedure by 4:00 p.m. on Friday, which helped meet the needs of epidemic prevention and control on the premise of ensuring products' safety and effectiveness.
A total of 57 registration certificates have been granted for disposable protective suits as well as 63, 171 and 364 such certificates for medical protective masks, surgical masks and disposable medical masks respectively across the country, according to Jiang.


