China's National Medical Products Administration has granted the world's first regulatory approval for an implantable brain-computer interface (BCI) device. This moves BCI technology — long the pinnacle of human-machine interaction — from lab research to clinical use.
BCI technology, first explored in the 1970s, creates direct brain-to-device communication.
"The fundamental principle is to establish an information pathway between the brain and external equipment that bypasses peripheral nerves or muscles," said Hong Bo, a professor at Tsinghua University's School of Biomedical Engineering.
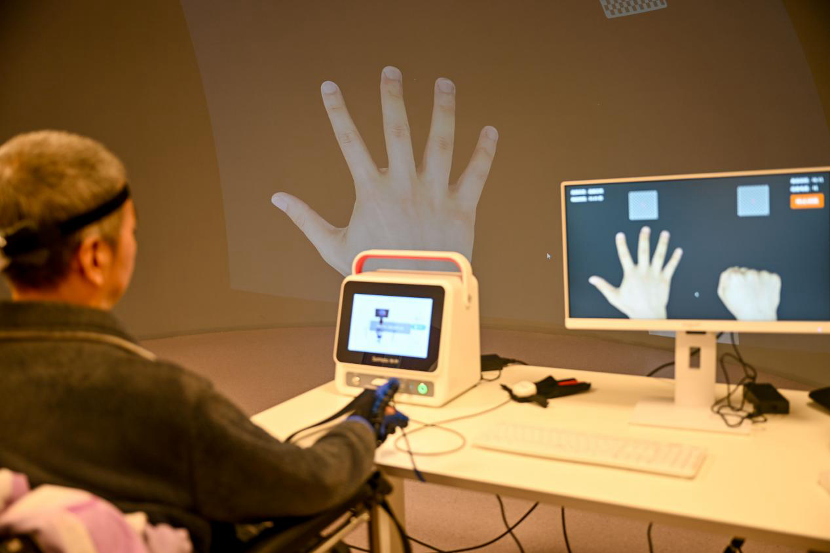
Patient rehabilitation training with brain-computer interface at Guizhou Hospital of Beijing Jishuitan Hospital, Guiyang, Guizhou Province. (Photo: People's Daily)
It collects neural signals from brain activity and converts them into commands, allowing thought control of external devices.
The newly approved implantable BCI system, or NEO system, approved for hand motor function, was developed by Neuracle Technology (Shanghai) Co., Ltd. (Neuracle) and Tsinghua University's School of Biomedical Engineering.
"For quadriplegic patients with cervical spinal injuries who can't grasp objects, we implant a coin-sized device outside the dura mater using minimal invasive surgery," said Wang Yujing, Neuracle product director.
"The system decodes brain signals in real time, enabling them to control a pneumatic glove with their thoughts to grasp objects or drink water," she added.
"Implantable BCIs pose the greatest technical challenges," Wang noted. Implanting electrodes into the cerebral cortex requires craniotomy, faces long-term tissue responses and demands reliable wireless transmission and safe power.
Earlier systems used wearable devices with visible wires. Mu-ming Poo, an academician at the Chinese Academy of Sciences and director of the Center for Excellence in Brain Science and Intelligence Technology, said recent advances in miniaturized chips, decoding algorithms and high-precision electrodes have enabled clinical transition.
China has completed dozens of clinical implantations. Trial data show that all patients improved their grasping ability, with some showing neural remodeling and additional functional recovery.
"The breakthrough lies in achieving stable brain signal acquisition through minimally invasive dura-outside implantation without neuron damage, plus accurate decoding of motor intent for grasping and drinking," said Mao Ying, president of Shanghai's Huashan Hospital (Fudan University).

A paraplegic patient grasps a water bottle using the NEO system in a clinical trial. (Photo provided by Tsinghua University)
In 2024, trial participant Dong — paralyzed by spinal injury — grasped a cup and drank independently using thought-controlled pneumatic gloves after training.
"These were impossible actions since his injury," Mao recalled. With rehab, Dong lifted dumbbells by mind control and hand-wrote "thank you" characters.
"Seeing BCI improve real lives was profoundly moving," Mao said.
Recently, Beijing Tiantan Hospital and Xuanwu Hospital (Capital Medical University), implanted "Beinao No. 1" systems. Feather-light 128-channel electrode arrays were placed in hand-movement brain regions, thereby aiding motor function in patients with spinal injuries.
"BCIs deliver real improvements for spinal injuries and stroke patients," said Li Yuan, business development director at Beijing startup NeuCyber Neuro Tech.
BCIs integrate materials science, chips, algorithms and rehab tech. An expert said China has built a full industrial chain, though it is still in its early stages. This approval will drive upstream components and downstream applications.
In July 2025, China issued BCI industry development guidelines, urging breakthroughs in core hardware/software, high-performance products and real-world applications. Beijing, Shanghai, Shandong and others introduced supportive policies.
China has 3.7 million spinal injury patients, with 90,000 new cases yearly, creating vast BCI potential.
Some predict that by 2027, China's BCI market will hit 5.58 billion yuan (about $818.46 million), growing 20 percent annually as medical rehab leads, with broader applications emerging.
"With strong policy support, continued technological development, declining costs, growing market awareness and improving regulatory frameworks, BCIs are expected to achieve larger-scale commercial application within the next three to five years, bringing tangible benefits to more people," Li said.


