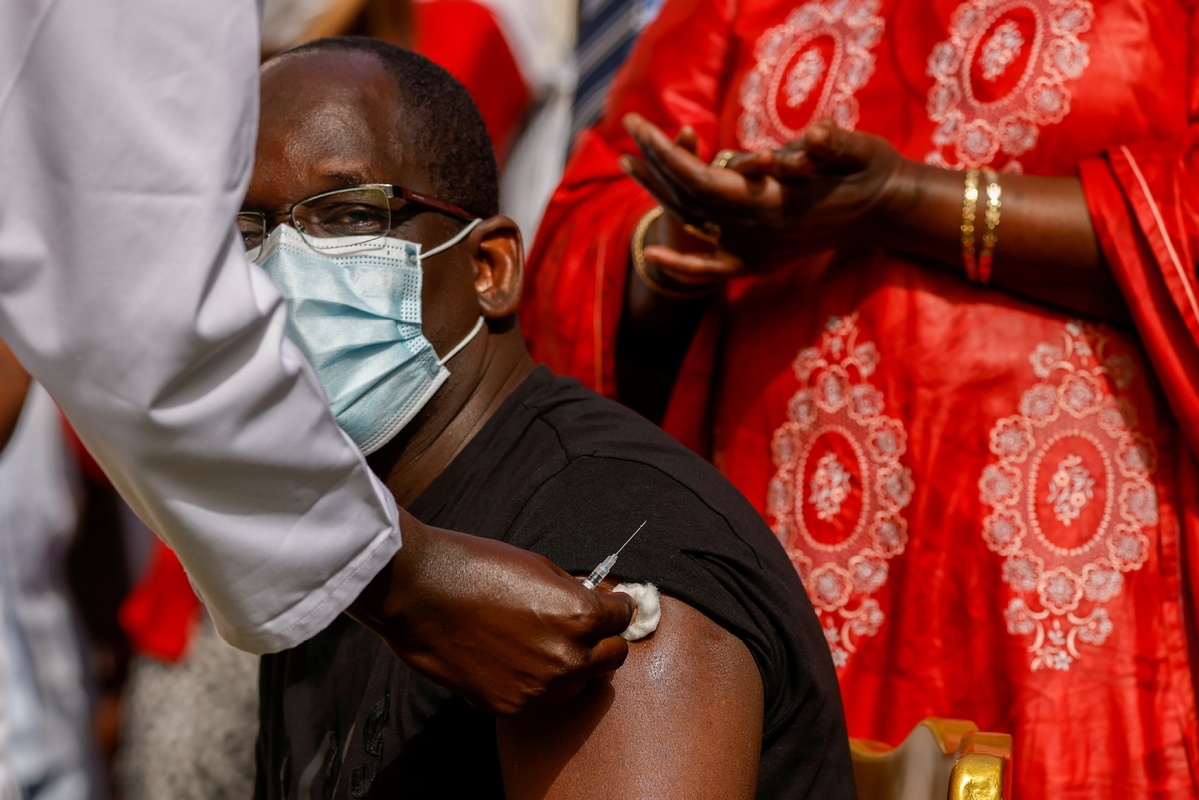
Health minister Abdoulaye Diouf Sarr receives a dose of the COVID-19 vaccine in Dakar, Senegal, Feb 23, 2021. (Photo: Agencies)
DAKAR - Senegalese Minister of Health and Social Action Abdoulaye Diouf Sarr, the first person inoculated with China's Sinopharm vaccine in Senegal, said Tuesday that he is doing well after taking the jab on Feb 23.
"I am doing well and I hope that immunity is setting in. In a couple of weeks, we will take the second dose," he said, assuring that the second dose will be guaranteed to all those that have been inoculated with the first dose.
Speaking to the 20H Journal on Senegalese public television RTS, the minister said that Senegal is the fourth African country in terms of inoculation number during the campaign of vaccination against COVID-19, thanks to the first batch of vaccines provided by Chinese pharmaceutical firm Sinopharm.
"We have vaccinated more than 35,000 Senegalese," said the minister, adding that the number of people vaccinated against COVID-19 in the country may reach around 40,000 as of Wednesday.
He also confirmed that his country will receive 324,000 doses of the COVID-19 vaccines from the COVAX initiative on Wednesday.
"It's encouraging. We must recognize that Senegal with this second batch, will enter an even more aggressive phase in terms of vaccination," he said.
Senega, which reported its first confirmed COVID-19 case on March 2, 2020, has so far recorded 34,832 positive cases, including 29,402 recoveries and 888 deaths.


